Mechanical Cooling
34 Direct Expansion Air Conditioning Systems
Click play on the following audio player to listen along as you read this section.
Direct expansion, or DX cooling, uses the principles of thermodynamics to transfer heat from one area to another through the evaporation and condensation of a refrigerant, which serves as the medium through which heat is captured and removed from one area and released in another.
Air conditioners use this mechanism to move heat from inside a room to outside, meaning the air conditioning system must have a component installed inside the room or area to collect thermal energy, and another component outside the room to release that thermal energy to the environment.
Refrigerators and freezers use DX cooling to remove thermal energy from inside the freezer unit to the outside. They utilize the same components as an air conditioning system, though on a smaller scale and creating much colder temperatures.
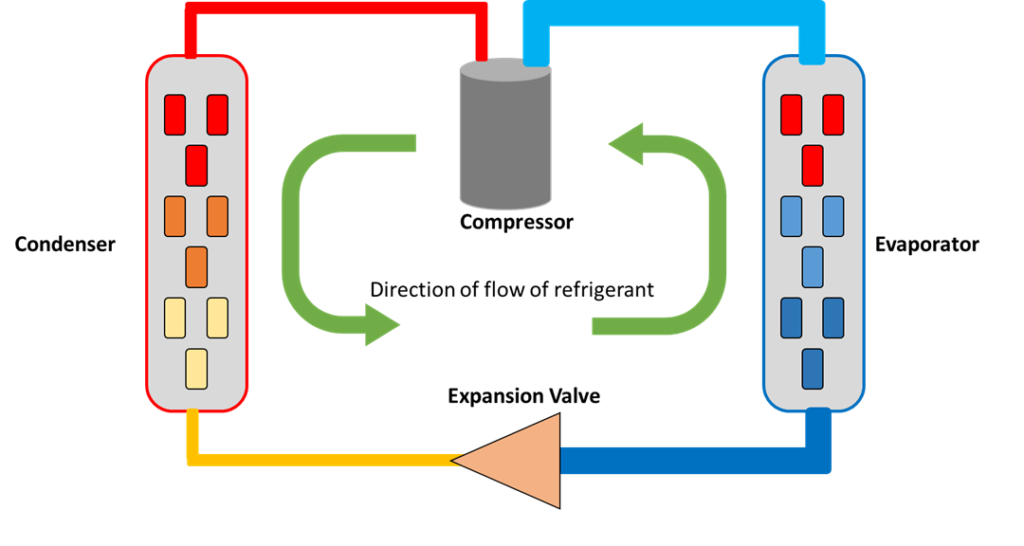
There are four main components of any DX cooling system, plus a heat absorbent medium, often called the refrigerant, that regulate the transference of heat energy by creating the temperature and pressure differentials required for DX cooling:
- The refrigerant, which is the medium that flows through the system, collecting and dissipating heat in different areas;
- The compressor, which is an electrical motor load and supplies the energy to drive the refrigerant through the system;
- The evaporator, which collects heat from the area, and facilitates the boiling of the refrigerant;
- The condenser, which dissipates heat into the ambient environment by allowing the refrigerant to return to a liquid state;
- The expansion valve, which acts as a regulator between the high and low pressure side of the system and allows for the drop in pressure and temperature necessary to facilitate DX cooling.
The whole system forms a closed loop, and is powered by the motor driven compressor. This device supplies the energy that drives the cooling system, and is usually a single-phase electrical motor load.
The compressor drives the next most critical component, the refrigerant, throughout the system. The refrigerant must have the property of boiling below room temperature. There are several varieties of refrigerant in use today, each with their own unique properties, but all share the common trait of evaporating at low temperatures. It is the property of changing phase, i.e. boiling, at low temperatures, that allows the refrigerant to maximize its ability to absorb thermal energy from a room or area.
The two components which facilitate the transfer of heat to or from the refrigerant are the evaporator located in the area to be cooled, and the condenser, located where heat may be dissipated, commonly outside of the building. The majority of the heat energy is dissipated in the condenser, and in order for heat transfer to occur, the air surrounding the condenser coil must be at a lower temperature than the refrigerant. This is why the compressor raises the temperature and pressure of the refrigerant, thus ensuring that there is sufficient heat gradient between the outside air and the refrigerant.
Between the condenser and the evaporator is the expansion valve, which regulates the pressure of the refrigerant. By allowing the refrigerant to expand, we increase the volume of space the gas can occupy, and thus lower the number of collisions that the atoms will have. This results in a lowering of kinetic thermal energy.
[latex]\text{Temperature}=\dfrac{\text{Pressure}}{\text{Volume}}[/latex]
Since the expansion valve lowers the pressure of the gas by allowing it to expand into the larger volume of the intake pipe feeding the evaporator, the refrigerant experiences a decrease in temperature. This is how we can actively make something colder than room temperature.
Refrigerants are designed to have a boiling point low enough to evaporate and turn into a gas at room temperature. For heat to transfer there has to be a temperature difference between the room and the refrigerant. If the cold refrigerant now flows through the evaporator coils, and air blown across the evaporator coils is at a higher ambient temperature, the colder refrigerant will absorb the heat, or thermal energy from the air, which has the same effect as cooling that air.
By constantly cycling additional cold refrigerant through the room, heat can be steadily removed from the desired area, and dissipated outside the building.
A mechanical cooling system that uses the evaporation and condensation of a refrigerant to remove heat from an area.
The branch of science that studies the relations between heat and other forms of energy.
A phase change where the entire material converts from a liquid to a gas.
A phase change where the entire material converts from a gas to a liquid.
A material used in DX cooling systems that boils below room temperature, and is used to transfer thermal energy from one point to another.
The process of cooling a room below the ambient temperature.
A measure of the average molecular kinetic energy of a system on a definite scale. Measured in degrees Fahrenheit, Celsius or Kelvin.
The force acted upon an object or medium by the surrounding environment. Often measured in PSI or Pascal’s.
A motor-driven component that pushes a fluid medium through a system.
A component which allows a refrigerant to rapidly boil and absorb thermal energy from a room or area. Will incorporate coils to maximize surface area to volume ratio.
A component which allows a refrigerant to rapidly condense and dissipate thermal energy into the environment. Will incorporate coils to maximize surface area to volume ratio.
A valve which allows a refrigerant to expand into a larger volume.
The amount of space that an object or medium takes up. Measured in cubic units (cm3) or liters.

