Mechanical Cooling
33 Phase Changes
Click play on the following audio player to listen along as you read this section.
https://media.bccampus.ca/id/0_vfamt5u1?width=608&height=80&playerId=23449753
We are relatively familiar with the three main forms of matter: solids, liquids and gasses. Since cooling systems make use of the changing properties of a refrigerant as it goes from liquid to gas form, it is useful to understand the relationships between temperature and heat energy in a system.
Solids are defined as having a low heat-energy level, meaning the atoms are locked in place and the object has a fixed volume and shape. For example a block of ice is water in its solid form.
As ice melts it becomes the liquid water that we are all familiar with. Its atoms are free to flow about, and it has a fixed volume, but will spread out to fill whatever container it is in, meaning its shape is not fixed.
As water is heated, it eventually boils and becomes a gas. In this state, its atoms are free to move about, and it has no fixed shape or volume. This is its highest energy level.
The relationship between the temperature of a medium versus the amount of heat energy added or removed from the environment is shown below.
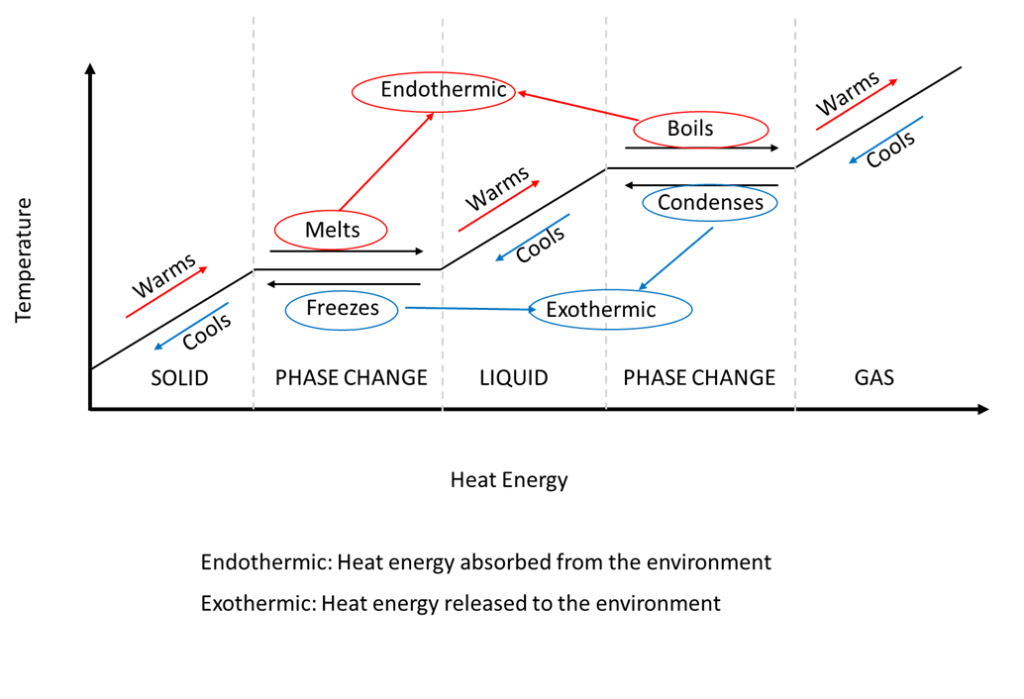
It is important to note the flat portions of the graph labelled “Phase Change” are horizontal with respect to the temperature axis. This means that a significant amount of heat energy must be either added or removed from the surrounding environment in order to change a material from a solid to a liquid, or a liquid to a gas. This heat energy is sometimes referred to as latent heat.
Only once the material has completely changed its phase will the temperature continue to rise. Once the materials temperature starts to rise, the energy which is now added (or removed) from the system is called sensible heat, because a person could sense, or feel the difference in temperature.
Endothermic reactions such as melting or boiling take energy from the surrounding environment and use it to change the phase of the material in the cooling system.
Exothermic reactions such as freezing or condensing release energy from the system back into the environment.
Direct Expansion cooling systems use the boiling and condensing properties of a refrigerant to rapidly remove thermal energy from an area where it is undesirable, and dissipate it elsewhere.
A material at its lowest energy state which will maintain a fixed volume and shape, such as a block of ice.
A material in its moderate energy state which will maintain a fixed volume but will move to fill any shape it is contained in, such as a glass of water.
A material in its highest energy state which will expand to fill the full volume or shape of the container, such as steam.
A material used in DX cooling systems that boils below room temperature, and is used to transfer thermal energy from one point to another.
A measure of the average molecular kinetic energy of a system on a definite scale. Measured in degrees Fahrenheit, Celsius or Kelvin.
The amount of energy added to or removed from a system required to change the temperature or phase of medium.
A phase change where the entire material converts from a solid to a liquid.
A phase change where the entire material converts from a liquid to a gas.
The thermal energy required to cause a medium to undergo a phase change.
The thermal energy required to raise or lower the temperature of a medium.
Heat energy absorbed from the environment, and added to the heating/cooling system
Heat energy released to the environment from the heating/cooling system.
A phase change where the entire material converts from a liquid to a solid.
A phase change where the entire material converts from a gas to a liquid.
A mechanical cooling system that uses the evaporation and condensation of a refrigerant to remove heat from an area.

