Chapter 5 Weathering and Soil
5.6 Weathering and Climate Change
Earth has two important carbon cycles. One is the biological one, wherein living organisms—mostly plants—consume carbon dioxide from the atmosphere to make their tissues. After they die almost all of that carbon is released back into the atmosphere when they decay over a period of years or decades. A small proportion of this biological-cycle carbon becomes buried in sedimentary rocks: during the slow formation of coal, as tiny fragments and molecules in organic-rich shale, and as the shells and other parts of marine organisms in limestone. This then becomes part of the geological carbon cycle, a cycle that actually involves a majority of Earth’s carbon, but one that operates very slowly.
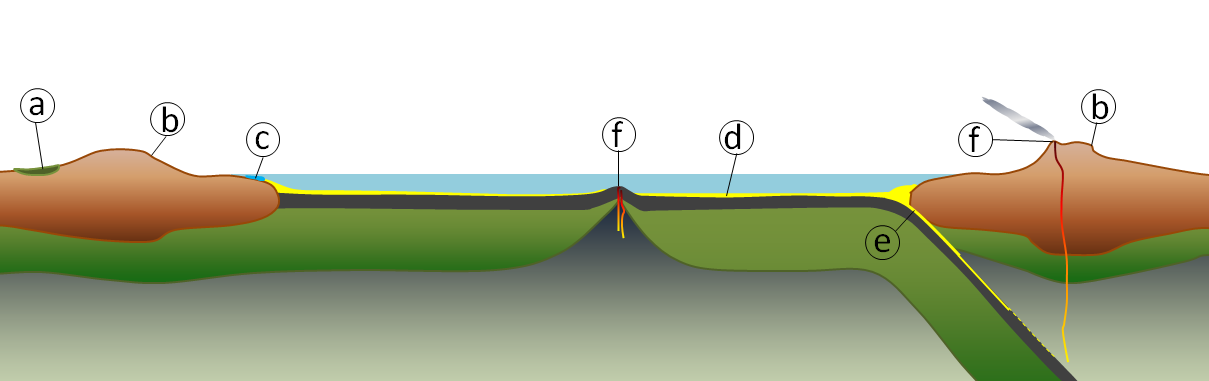
The geological carbon cycle is shown diagrammatically in Figure 5.6.1. The various steps in the process (not necessarily in this order) are as follows (the letters are shown on Figure 5.6.1):
a) Organic matter from plants is stored in lake sediments, peat, and permafrost for up to tens of thousands of years, and some may be buried deeper to form coal that can be stored for tens of millions of years.
b) Weathering of silicate minerals converts atmospheric carbon dioxide to dissolved bicarbonate, which is stored in the oceans for thousands to tens of thousands of years.
c) Dissolved carbon is converted by marine organisms to calcite, which is stored in carbonate rocks for hundreds of millions of years.
d) Organic and inorganic carbon compounds are stored in sediments for tens to hundreds of millions of years; some end up in petroleum deposits.
e) Carbon-bearing sediments are transferred to the mantle, where the carbon may be stored for tens of millions to billions of years.
f) During volcanic eruptions, carbon dioxide is released back to the atmosphere, where it is stored for years to decades.
During much of Earth’s history, the geological carbon cycle has been balanced, with carbon being released by volcanism at approximately the same rate that it is stored by the other processes. Under these conditions, the climate remains relatively stable.
During some periods of Earth’s history, that balance has been upset. This can happen during prolonged periods of greater than average volcanism. One example is the eruption of the Siberian Traps at around 250 Ma, which appears to have led to strong climate warming over a few million years because of the slow but steady input of extra volcanic CO2 into the atmosphere.
A carbon imbalance is also associated with significant mountain-building events. For example, the Himalayan Range was formed between about 40 and 10 Ma and over that time period—and still today—the rate of weathering on Earth has been enhanced because those mountains are so high and steep and the range is so extensive. The weathering of these rocks—most importantly the hydrolysis of feldspar—has resulted in consumption of atmospheric carbon dioxide and transfer of the carbon to the oceans and to ocean-floor carbonate minerals. The steady drop in carbon dioxide levels over the past 40 million years, which led to the Pleistocene glaciations, is partly attributable to the formation of the Himalayan Range.
A non-geological form of carbon-cycle imbalance is happening today on a very rapid time scale. We are in the process of extracting vast volumes of fossil fuels (coal, oil, and gas) that was stored in rocks over the past several hundred million years, and then converting these fuels to energy and carbon dioxide. By doing so, we are changing the climate faster than has ever happened in the past, and putting both ecosystems and our descendants at significant risk.
Media Attributions
- Figure 5.6.1: © Steven Earle. CC BY.

