Chapter 8. Intravenous Therapy
8.3 IV Fluids, IV Tubing, and Assessment of an IV System
Patients are prescribed an IV solution (fluids) based on their electrolyte and fluid volume status. IV fluids are commonly categorized as colloids and crystalloids. Colloid solutions contain large molecules that cannot pass through semi-permeable membranes and are used to expand intravascular volume by drawing fluid from extravascular space via high osmotic pressure. Examples of colloid solutions are albumin, dextrans, and hydroxyethyl starches (Crawford & Harris, 2011). Crystalloid solutions contain solutes such as electrolytes or dextrose, which are easily mixed and dissolvable in solution. Crystalloids contain small molecules that flow easily across semi-permeable membranes, which allows for transfer from the bloodstream into the cells and tissues (Crawford & Harris, 2011). They may increase fluid volume in interstitial and intravascular space. Examples of crystalloid solutions are isotonic, hypotonic, and hypertonic solutions.
Isotonic solutions have an osomolality of 250 to 375 mOsm/L. Isotonic solutions have the same osmotic pressure as plasma, creating constant pressure inside and outside the cells, which causes the cells to remain the same (they will not shrink or swell) and does not cause any fluid shifts within compartments. Isotonic solutions are useful to increase intravascular volume, and are utilized to treat vomiting, diarrhea, shock, and metabolic acidosis, and for resuscitation purposes and the administration of blood and blood products. Examples of isotonic solutions include normal saline (0.9% sodium chloride), lactated Ringer’s solution, 5% dextrose in water (D5W), and Ringer’s solution. It is important to monitor patients receiving isotonic solutions for fluid volume overload (hypervolemia) (Crawford & Harris, 2011).
Hypotonic solutions have a lower concentration, or tonicity, of solutes and have an osomolality equal to or less than 250 mOsm/L. The infusion of hypotonic solutions lowers the osmolality within the vascular space and causes fluid to shift to the intracellular and interstitial space. Cells will swell but may also delete fluid within the vascular space. Examples of hypotonic solutions include 0.45% sodium chloride, 0.33% sodium chloride, 2.5% dextrose in water, and 0.2% sodium chloride. Monitor for hypovolemia and hypotension related to fluid shifting out of the vascular space, and do not administer to patients with increased intracranial pressure (ICP), as it may exacerbate cerebral edema. Use cautiously in patients with burns, liver failure, and traumas (Crawford & Harris, 2011).
Hypertonic solutions have a higher concentration, or tonicity, of solutes and have an osomolality equal to or greater than 375 mOsm/L. The osmotic pressure gradient draws water out of the intracellular space into the extracellular space. Examples of hypertonic solutions include D5W and 0.45% sodium chloride, D10W, and 3% sodium chloride. Hypertonic solutions may cause intravascular fluid volume overload and pulmonary edema, and they should not be used for an extended period of time. Hypertonic solutions should not be used in patients with heart or renal disease who are dehydrated (Crawford & Harris, 2011).
Although all IV fluids must be administered carefully, hypertonic solutions are additionally risky.
An order for IV fluids may be continuous or as a bolus, depending on the needs of the patient. IV solutions are available in 25 ml to 1000 ml bags. The frequency, duration, amount, and additives to solution must be ordered by a physician or nurse practitioner; for example, an order may be “give NS at 125 ml/hr.”
The most common types of solutions include normal saline (NS) and D5W. Patients may also have medications, such as potassium chloride, thiamine, and multivitamins, added to IV solutions. To discontinue an IV infusion, an order must be obtained from the physician or nurse practitioner (Perry et al., 2014).
IV Administration Equipment
When a peripheral vein has a cannula inserted, an extension tubing is connected to the hub on the cannula and flushed with normal saline to maintain patency of the cannula. Most peripheral intravenous cannulas will have extension tubing, a short, 20 cm tube with a positive fluid displacement/positive pressure cap attached to the hub of the cannula for ease of access and to decrease manipulation of the catheter hub (Vancouver Coastal Health, 2008). The extension tubing must be changed each time the peripheral catheter is changed. When the peripheral cannula is not in use, the extension tubing attached to the cannula is called a saline lock.
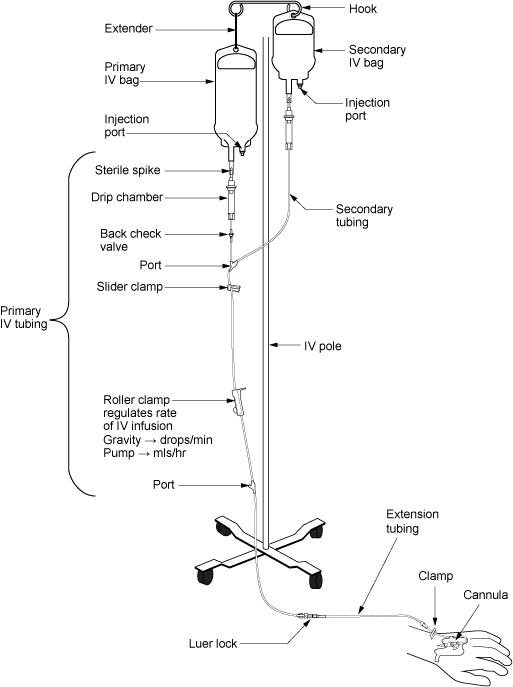
Intravenous fluids are administered through thin, flexible plastic tubing called an infusion set or primary infusion tubing/administration set (Perry et al., 2014). The infusion tubing/administration set connects to the bag of IV solution. Primary IV tubing is either a macro-drip solution administration set that delivers 10, 15, or 20 gtts/ml, or a micro-drip set that delivers 60 drops/ml. Macro-drip sets are used for routine primary infusions. Micro-drip IV tubing is used mostly in pediatric or neonatal care, when small amounts of fluids are to be administered over a long period of time (Perry et al., 2014). The drop factor can be located on the packaging of the IV tubing.
Primary IV tubing is used to infuse continuous or intermittent fluids or medication. It consists of the following parts:
- Backcheck valve: Prevents fluid or medication from travelling up the IV
- Access ports: Used to infuse secondary medications and give IV push medications
- Roller clamp: Used to regulate the speed of, or to stop or start, a gravity infusion
- Secondary IV tubing: Shorter in length than primary tubing, with no access ports or backcheck valve; when connected to a primary line via an access port, used to infuse intermittent medications or fluids. A secondary tubing administration set is used for secondary IV medication.
IV solution bags should have the date, time, and initials of the health care provider marked on them to be valid. Add-on devices (e.g., extension tubing or dead-enders) should be changed every 96 hours, if contaminated when administration set is replaced, or as per agency policy. Intravenous solution and IV tubing should be changed if:
- IV tubing is disconnected or becomes contaminated by touching a non-sterile surface
- Less than 100 ml is left in the IV solution bag
- Cloudiness or precipitate is found in the IV solution
- Equipment (date and time) is outdated
- IV solution is outdated (24 hours since opened)
Primary and secondary administration sets (see Figure 8.4) should be changed regularly to minimize risk and prevent infection (CDC, 2011; Fraser Health Authority, 2014). Change IV tubing according to agency policy. Table 8.5 lists the frequency of IV tubing change.
Safety considerations:
|
|||
Frequency of IV Tubing Change |
Type of IV Tubing and Solution |
||
| Every 72 -96 hours | Primary tubing with hypotonic, isotonic, or hypertonic continuous solution, when insertion site is changed, or when indicated by the type of solution or medication being administered. | ||
| Every 24 hours | Secondary or intermittent IV solution or medication. Rationale: When an intermittent infusion is repeatedly disconnected and reconnected for infusion, there is increased risk of contamination at the catheter hub, needleless connector, and the male Luer end of the administration set, potentially increasing risk for CR-BSI. | ||
| Every 24 hours | Infusions containing fat emulsions (IV solutions combined with glucose and amino acids infused separately or in a 3 in 1 admixture). Example: Total parenteral nutrition (TPN). | ||
| 4 hours or 4 units, whichever comes first, or between products | Blood and blood products | ||
| Data source: CDC, 2011 | |||
Infusing IV Fluids by Gravity or an Electronic Infusion Pump (EID)
To ensure therapeutic effectiveness of IV fluids, a constant, even flow is necessary to prevent complications from too much or too little fluid. A physician must order a rate of infusion for IV fluids or for medications. The rate of infusion for medications (given via a secondary or primary infusion) can be found in the Parenteral Drug Therapy Manual (PDTM). If an order for IV fluids is “to keep vein open” (TKVO), the minimum flow rate is 20 to 50 ml per hour, or according to physician’s orders (Fraser Heath Authority, 2014).
A health care provider is responsible for regulating and monitoring the amount of IV fluids being infused. IV fluid rates are regulated in one of two ways:
- Gravity. The health care provider regulates the infusion rate by using a clamp on the IV tubing, which can either speed up or slow down the flow of IV fluids. An IV flow rate for gravity is calculated in gtts/min.
- Electronic infusion device (EID) (see Figure 8.5). The infusion rate is regulated by an electronic pump to deliver the fluids at the correct rate and volume. All IV pumps regulate the rate of fluids in ml/hr. An IV pump (EID) is used for many types of patients, solutions, and medications (Vancouver Coastal Health, 2008).
An IV pump must be used for:
- All CVC devices
- All opioid infusions (use a patient-controlled analgesia)
- All pediatric patients
- All medication as described in the PDTM
- Infusion rates below 60 ml/hr
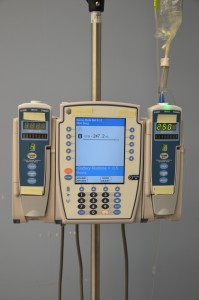
Figure 8.5 Electronic infusion device (EID)
To calculate the drops per minute for an infusion by gravity, follow the steps in Table 8.6.
When an infusion is by gravity, there are several factors that may alter the flow/infusion rate (Fulcher & Frazier, 2007). In addition to regulating the flow rate, assess the IV system to ensure these factors are not increasing or decreasing the flow of the IV solution. These factors are listed in Table 8.7.
|
Factors |
Additional Information |
||
| Tube occlusion | May occur if the tubing is kinked or bent. Tubing may become kinked if caught under the patient or on equipment, such as beds and bed rails. | ||
| Vein spasms | Irritating or chilled fluids (fluids stored in the fridge) may cause a reflex action that causes the vein to go into spasm at or near the intravenous infusion site. If fluids or medications are chilled, bring to room temperature prior to infusion. | ||
| Height of the fluid container | The IV tubing drip chamber should be approximately 3 feet above IV insertion site. | ||
| Location/position of IV cannula | If the cannula is located in an area of flexion (bend of an arm), the IV flow may be interrupted when the patient moves around. To avoid this issue, replace IV cannula. | ||
| Infiltration or extravasation | If the cannula punctures the vein, the fluid will leak into the surrounding tissue and slow or stop the flow, and swelling will develop. | ||
| Accidental touching/bumping of the control clamp or raising arm above heart level | Instruct the patient not to touch the roller clamp and to take care not to bump the clamp, as this may accidentally change the flow rate. Instruct patient to keep hand/arm below heart level; an elevated hand/arm will slow or stop an infusion running by gravity. | ||
| Needle or cannula gauge/diameter | The smaller the needle or cannula, the slower the fluid will flow. | ||
| Data source: Fulcher & Frazier, 2007; Perry et al., 2014 | |||
Assessing an IV System
All patients with IV fluid therapy (PIV and CVC) are at risk for developing IV-related complications. The assessment of an IV system (including the IV site, tubing, rate, and solution) (see Figure 8.6) often depends on what is being infused, the patient’s age and medical condition, type of IV therapy (PIV or CVC), and agency policy. Generally, an IV system should be assessed as described in Checklist 65.
Disclaimer: Always review and follow your hospital policy regarding this specific skill. |
||||
Safety considerations:
|
||||
Steps |
Additional Information |
|||
| 1. Perform hand hygiene. | This step reduces the transmission of microorganisms. | |||
| 2. Introduce yourself and explain the purpose of the assessment. | This builds trust with patient and allows time for the patient to ask questions. | |||
| 3. Confirm patient ID using two patient identifiers (e.g., name and date of birth), and compare the MAR printout with the patient’s wristband. | This step ensures you have the correct patient and complies with agency standard for patient identification.
 |
|||
| 4. Apply non-sterile gloves (optional). | This reduces the transmission of microorganisms. | |||
| 5. Assess the IV insertion site and transparent dressing on IV site. | Check IV insertion site for signs and symptoms of phlebitis or infection. Check for fluid leaking, redness, pain, tenderness, and swelling. IV site should be free from pain, tenderness, redness, or swelling.
Ensure patient is informed to alert the health care provider if they experience pain or notice swelling or redness at the IV site. If patient is unable to report pain at IV site, more frequent checks are required. |
|||
| 6. Inspect the patient’s arm for streaking or venous cords; assess skin temperature. | Assess complications on hand and arm for signs and symptoms of phlebitis and infiltration/extravasation. | |||
| 7. Assess IV tubing for kinks or bends. | Kinks or bends in tubing may decrease or stop the flow of IV fluids. Ensure tubing is not caught on equipment or side rails on bed.
Tubing should be properly labelled with date and time. |
|||
| 8. Check the rate of infusion on the primary and secondary IV tubing. Verify infusion rate in physician orders or medication administration record (MAR). | If IV solution is on gravity, calculate and count the drip rate for one minute.
If solution is on an IV pump, ensure the rate is correct and all clamps are open as per agency protocol. If secondary IV medication is infusing, ensure clamp on secondary IV tubing is open. The EID is unable to distinguish if the primary bag or secondary bag is infusing. |
|||
| 9. Assess the type of solution and label it on bag. Check volume of solution in bag. | IV solutions become outdated every 24 hours.
Ensure the correct solution is given. If 100 ml of solution or less is left in the bag, change the IV solution and document on in-and-out sheet. If an IV pump is used, ensure it is plugged into an outlet. Ideally, the IV solution should be 90 cm above patient heart level. |
|||
| 10. Assist patient into comfortable position, place call bell in reach, and put up side rails on bed as per agency policy. | These precautions prevent injury to the patient. | |||
| 11. Perform hand hygiene. | This step prevents the spread of microorganisms. | |||
| 12. Document procedure and findings as per agency policy. | Timely and accurate documentation promotes patient safety. | |||
| Data source: Fulcher & Frazier, 2007; Perry et al., 2014 | ||||
Critical Thinking Exercises
- What are the signs and symptoms of phlebitis?
- What types of patients should not receive hypotonic IV solutions?